ISO 13485 Medical Devices Quality Management System - Training Courses
What is ISO 13485?
As an international standard, ISO 13485 determines the requirements for the medical devices industry. This standard was established to be used by companies during the life cycle of medical devices, from the production to post-production, including decommission and disposal.
However, ISO 13485 can also be operated by other parties such as certification bodies which can assist in the certification processes. ISO 13485 serves as a tool for organizations to develop and retain their processes efficiently. Organizations that are striving for continuous improvement will benefit from this standard by being able to provide safe and competent medical devices and achieve the trust of customers.
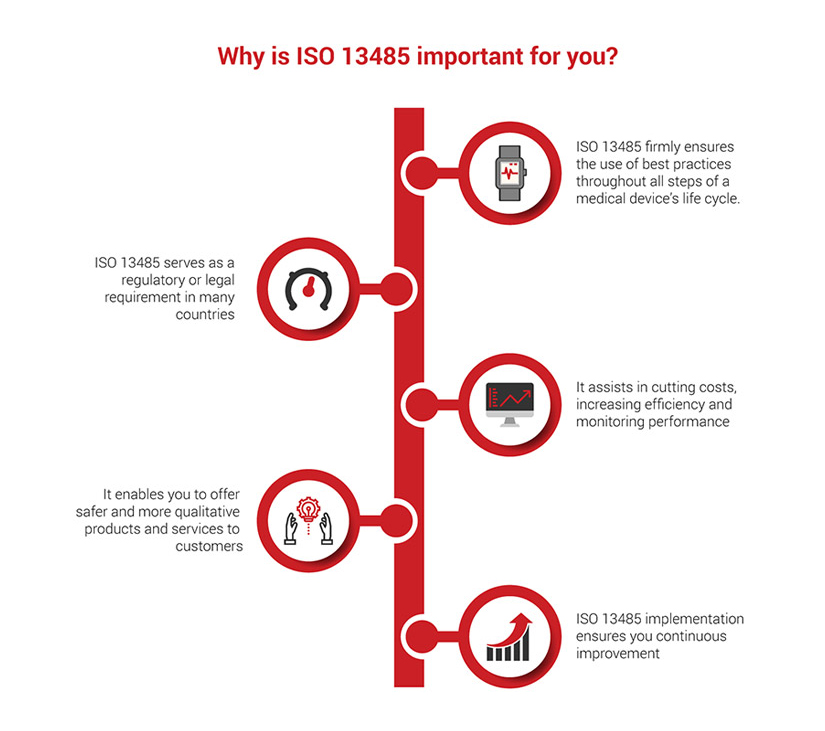
Why is Medical Devices Quality Management System important for you?
Being an ISO 13485 certified professional imposes your determination to offer qualitative products and services to your company and/or clients. Attaining the necessary knowledge and skills to operate an ISO 13485 framework demonstrates your commitment to helping your company ensure continuous improvement and better work processes.
It also indicates that you understand the importance of the safety and performance of medical devices and how a medical devices quality management system can assure just that, consequently leading to customer satisfaction. Similarly, it can introduce you to new opportunities, such as working for large companies that offer quality products and services. These companies will value your knowledge and expertise on this standard, while enabling you to maximize your earning potential.
Benefits of ISO 13485 Medical Devices Quality Management System
By becoming an ISO 13485 certified professional, you will be able to:
- Increase the number of job opportunities
- Create new business connections
- Gain competitive advantage
- Increase effectiveness and lower costs
- Offer expertise on improving work processes
- Offer safer and more efficient medical devices
- Increase customer satisfaction
How do I get started with ISO 13485 Training?
If you are willing to take the challenge of obtaining an ISO 13485 certification, our experts will ensure a qualitative experience where your needs will be met, and you will become part of our global network.
Contact us to start with the first step
PECB Certified ISO 13485 training courses available
Acquire knowledge on Medical Devices Quality Management Systems through our PECB ISO 13485 training courses. Check below to find the training that suits you best:
